|
Whether the Big Rip turns out to be true or not, since the end of the 20th century, our cosmic future has grown even shorter than we thought it was. In that account, which I find particularly intuitive and evo devo, we have already seen between a third and half of our universe’s total lifespan (figure right). In that model, dark energy acceleration gets increasingly strong with time, and in a few billion years from now, even matter itself gets ripped apart and recycled. One particularly accelerative model for end of our universe is the Big Rip. So the end of our universe is not only obviously ahead, we now know that this ending is an accelerative process. For the last few billion years, our universe has begun to take itself apart at an accelerating rate, making ever more space for its heat to “dump” into. Since the discovery of dark energy in 1998, we’ve also known that our universe’s creation of entropy is accelerating. Unfortunately, everything must eventually die, inside our universe. Many scientists consider it the most fundamental law in physical theory. This insight is called the second law of thermodynamics, and the “second law” is one of the best-tested laws in all of science. It was a big bummer for us at the time to learn that our universe is inevitably heading toward “ heat death,” the loss of sources of useful energy, via a process of ever-increasing entropy. We’ve known since William Thompson in 1852 that our universe is running down in its energy potential. Image Credit: Jeremy Teaford, Vanderbilt University Is product- or reactant-favored at 25 ✬.The Big Rip Model of the Fate of Our Universe. 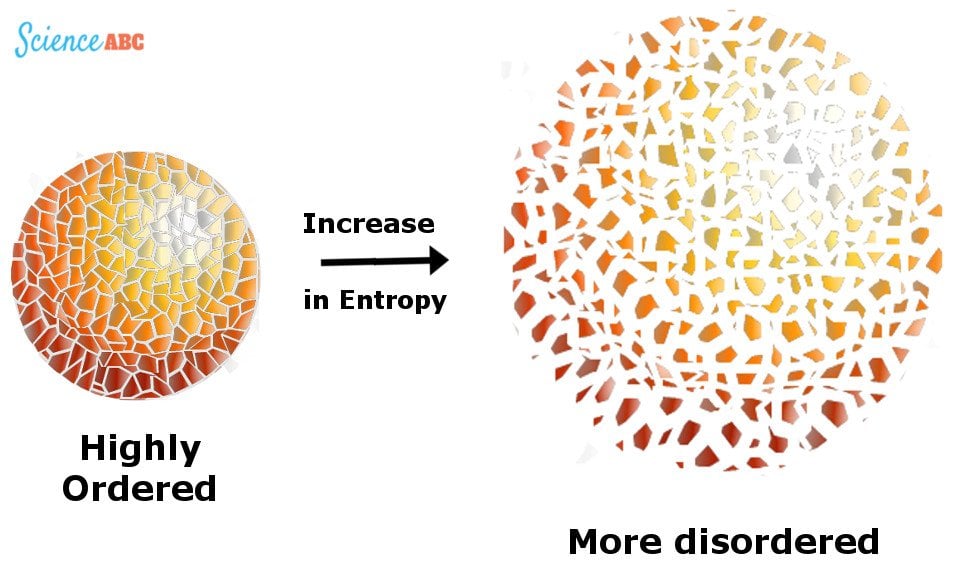
If it is less than zero, the reaction is reactant-favored. If it is greater than zero, the reaction is product-favored. Now that DS syst and DS surr are known, DS univ can be determined. DH syst can be calculated in the way described in the Thermochemical Equations module. So, q surr and DH syst must always have opposite signs, which is why DH syst is given a negative sign. Why does DH syst have a negative sign? Any heat lost by the system is gained by the surroundings, and conversely, any heat gained by the system is lost by the surroundings. Thus, equation (1) can be used to calculate DS surr: Since the surroundings are so much bigger than the system, its temperature is certain to stay constant. But, the only way the system affects the surroundings is by a transfer of heat. It may seem unlikely that the entropy change of the surroundings can be calculated just from what is known about the system. It can be calculated using absolute entropies as has been described on the previous page. 
Is product- or reactant-favored? The entropy change of the universe can beīroken up into two parts, the entropy change of the system and the entropyĭS syst, the entropy change of the system, represents the change in order of the molecules of the system, similar to what was discussed in Entropy 2. How can the Second Law of Thermodynamics be used to predict whether a reaction The formal statement of this fact is the Second Law of Thermodynamics: in any product-favored process the entropy of the universe increases. The situations described in the second and third pages of this tutorial illustrate the fact that product-favored reactions tend to increase disorder simply because they are much more likely to occur.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
